No. 33 Physical properties of R(+) alpha Lipoic acid by Complexation with γ-Cyclodextrin
Continued from No.21_RALA-CD Alpha Lipoic acid γ-cyclodextrin complex
What is alpha Lipoic acid?
Alpha Lipoic acid exists in nature and is an essential cofactore of mitochondrial enzyme complexes. Glucose taken into cells is changed into pyruvic acid by glycolytic pathway. Pyruvic acid is changed into acetyl CoA by pyruvate dehydrogenase complex (PDC), then acetyl CoA is oxidized at TCA cycle and electron transfer system followed by generation of ATP. Lipoic acid is essential for PDC and glucose metabolism. Continuous high blood glucose level causes generation of advanced glycation end products (AGEs). It is explained that AGEs cause the complications of diabetes mellitus and aging. Alpha Lipoic acid which can works for conversion from glucose to energy is now very popular as anti glycation materials.

Goal
The carbon atom at C6 of Alpha Lipolic acid is chiral and the molecule exists an two enantiomers R-(+)-Lipoic acid (RALA) and S-(-)-lipoic acid (SALA). Only the RALA exists in nature and is an essential cofactor of four mitochondrial enzyme complexes. Since RALA (mp. 46-49℃) is unstable compound, it is easily decomposed and generates sticky insoluble polymer. Therefore, RALA has not been commercially available, but only racemic compound (mp. 60-62℃).
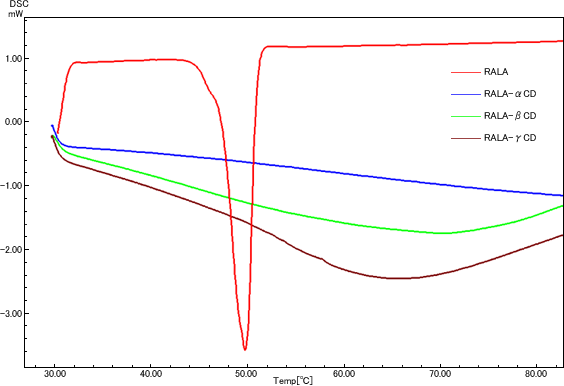
RALA-CD inclusion complexes were prepared, and their stability against heat and acid conditions were evaluated.

RALA-CD complex
CD complexation procedure has been applied to patent and NOT disclosed. (JP2010-172420)
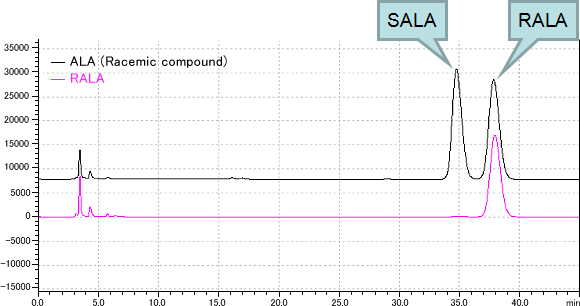
Analysis of RALA by HPLC
Unit : Integrated HPLC System LC-2010C (Shimadzu)
Column : CHIRALPAK AD-RH (Daicel) (4.6mm I.D. x 150mm)
Flow and temp. : 0.6mL/min, 25℃
Mobile phase : 25mM H3PO4:CH3CN=70:30
Detector : UV 215nm
Injection volume : 10μL
Retention time : 35min (SALA), 38min (RALA)
Chromatogram
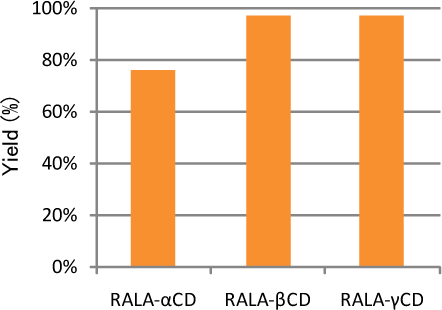
Efficiency of complexation process
Results
RALA contents in RALA-CD complexes which were made by mixing RALA and CD (1:1 mol ratio) were analyzed by HPLC.
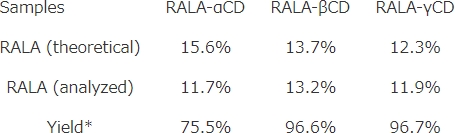
Yield (%) = RALA (analyzed) /RALA (theoretical)
Stabilizing effect of CDs on RALA
Residual ratio of RALA under heated condition
(Light shielding, 70℃, saturated humidity, 2h)
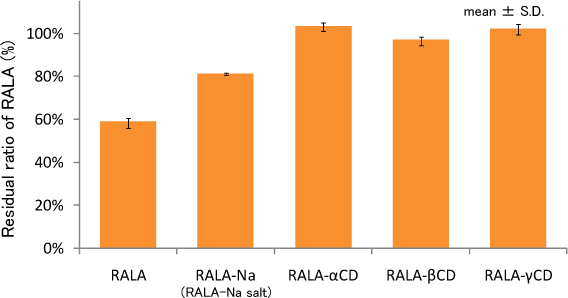
Each samples were treated under the test conditions (light shielding, 70℃ and saturated humidity) for 2 hours, then residual RALA content was analyzed. The results were shown as residual ratio of RALA against initial value.
Evaluation of RALA-γ-CD complex
Stability of RALA in water condition
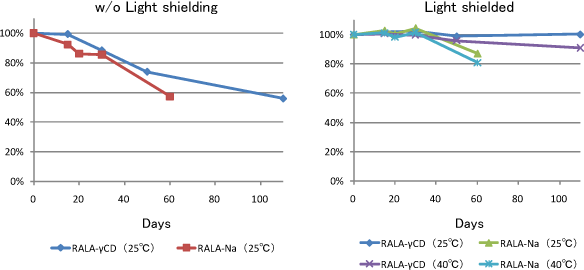
RALA contents in each samples were 0.5mg/mL.
Stability of RALA in Water (90℃)
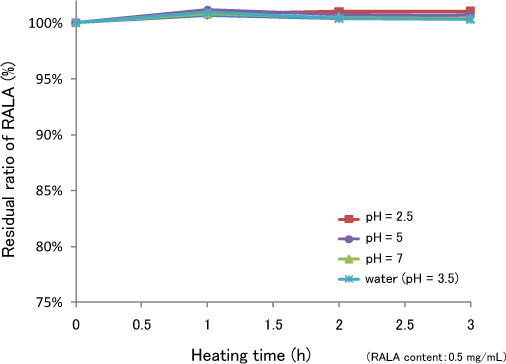
Stability of RALA in heated water

RALA didn't dispersed in water. RALA was polymerized under heated condition.
Taste masking effect by γ-CD
Sensory test
【Panelist】
Healthy adult aged 20's to 60's (Male 8, Female 7)
【Materials】
γ-CD, RALA-γ-CD , RALA-MCC*
(Both RALA-γ-CD and RALA-MCC contain 11% of RALA)
RALA-MCC:Physical mixture of RALA and crystalline cellulose.
【Method】
Panelist didn't know the content of test sample.
Panelist rinses the mouth then lick the test sample followed by evaluation its pungency in the mouth.
【Score】
No pungency = 0
1 (less pungency) to 10 (strong pungency)
【Screening and judge】
Two panelists who gave 1 score to γ-CD were eliminated.
Sum total of each sample were evaluated.
Results
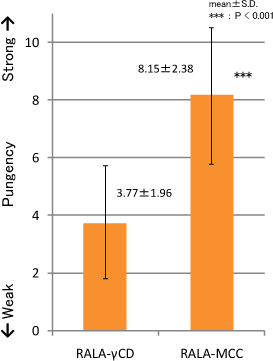
Pungent taste of RALA was decreased by γ-CD complexation.
Safety evaluation of RALA-γ-CD
Acute toxicity test
【試験動物】
5週齢のICR系雄雌マウスを約1週間の予備飼育を行い、異常のないことを確認した後、ポリカーボネート製ゲージに各5匹ずつを収容し、室温23℃±2℃、照明時間12時間/日に設定した飼育室にて飼育した。飼料及び飲料水は自由に摂取させた。
【試験液の調製】
検体を注射用水で懸濁し、100mg/mLの試験液を調製した。
【試験方法】
試験動物は投与前に約4時間絶食させた。体重を測定した後、試験群には試験液、対照群には注射用水をそれぞれ20mL/kg体重の投与容量で胃ゾンデを用いて強制単回投与した。
試験群:雄雌各5匹 検体投与量:2000mg/kg体重
対照群:雄雌各5匹 注射用水を投与
【結果】
14日間観察の結果、異常や死亡例は認められなかった。
Body weight transition
mean ± S.D. (g)
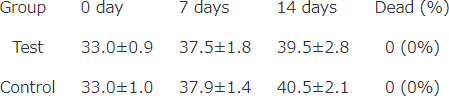
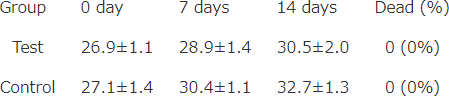
LD50 > 2000mg/kgBW (Mice)
(試験実施機関:(財)本食品分析センター)
Conclusion
- RALA can make inclusion complex by α-CD, β-CD and γ-CD.
- The stability of RALA was improved by making inclusion complex with CD.
- RALA-γ-CD complex was stable even in solution.
- The pungency taste of RALA was decreased by γ-CD.
- LD50 of RALA-γ-CD complex was > 2000mg/kgBW (mice).
↓
RALA-γ-CD complex is now available as functional food materials !
